FUEL MICROBIOLOGY – WHAT IS THE RISK OF INFECTING A TANK?
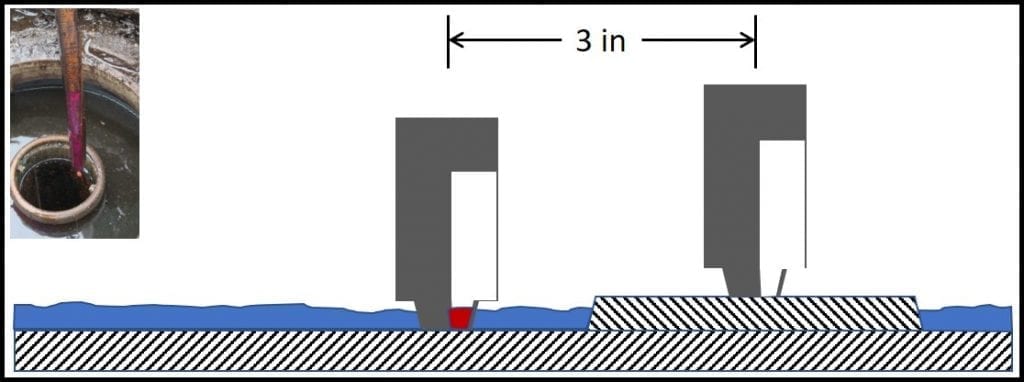
I recently received a question regarding the use of one tank-stick to measure multiple tanks. The question was: “if you stick a tank that is contaminated into the next tank, will it contaminate the second tank?” That is: can the microbial load carried over from UST to another, on a gauging stick, infect the second UST?
Given how much press there has been lately about how easy it is to spread disease through brief, hand contact with contaminated surfaces, this is an excellent question. I thought that others who read this blog might be interested in the issue.
Here’s my response to the question:
Interesting question!.
No doubt your are extrapolating from your general understanding of how diseases can be easily transmitted either by the traces we transfer from first contacting a contaminated surface and then eating a sandwich – thereby ingesting the microbes we just transferred from our hands to the sandwich. Or, perhaps a better example is how easily viral diseases are transferred by mosquitos. A fraction of a mL of mosquito saliva can transfer a sufficient number of viruses from an infected host to a new victim.
Anything is possible, but there are a number of factors that reduce the likelihood of a gauging stick being the primary vector for microbial contamination transmission among fuel tanks:
• If a technician is using water paste, they are likely to wipe down the stick between tanks.
• Fuel is volatile; evaporation after the stick is pulled from a tank is likely to desiccate (dry out) any microbes that adhered to the stick while it was in the tank. If they are not already in a dormant state, the microbes adhering to the stick won’t have had sufficient time to transform from the active (vegetative) to dormant state before the product evaporates. Even though diesel evaporates more slowly than gasoline, it acts as a desiccant (that’s why vegetative microbes are not found in fuel that doesn’t have dispersed water present).
• Fuel systems are more hostile environments than human bodies. In microbiology we have a concept of minimum infectious dose. Typically that minimum is in the thousands or millions of cells. If the number of cells transferred is below the minimum infectious dose, then the population will most likely die off rather than seed the development of a new population in an uninfected tank that is gauged after an infected tank has been gauged.
Also, consider volumes.
• Water: 100 ppm water in 10,000 gal fuel = 1 gal water. If only 10% of that dissolve/dispersed water settles out, that’s 0.1 gal/delivery. A tank receiving only 1 delivery/wk will accumulate >5 gal/year in free-water. My 10% dropout rate is based on daily deliveries, so it’s more likely (and common) to see closer to 300 gal/y.
• Air: this might be changing as newer vapor recovery and vent systems replace current systems, but the volume of air entering a tank equals the volume of fuel withdrawn. These systems do not scrub water, pollen or dust from the air. A USAF global fuel system survey completed about 10 years ago determined that the profile of microbes found in fuel tanks closely mimicked that found in the nearby air (the research team took air samples and tank bottom samples). The research team reported much closer relationships between what they found in the air near fuel tanks and what they found I fuel tanks, than between the fuel grade and microbial community profile. These results – of course – strongly supported the hypothesis that tank vents are a (if not the) major source of microbial contamination in fuel tanks.
The next time I’m in the field performing a microbial audit of fleet or retail sites, I’ll test sounding sticks before and after using them to measure bottoms-water and product. If I find that sounding sticks are indeed picking up significant microbial loads, I’ll report that at a D02.14 Fuel Microbiology meeting, and might even write a paper on the issue.